Throughout the first semester, our laboratory subteam has been hard at work at developing technical memos and completing research about potential reactions to use in future Chem-E-Cars. Below is a summary of the research and work they have completed throughout the term.
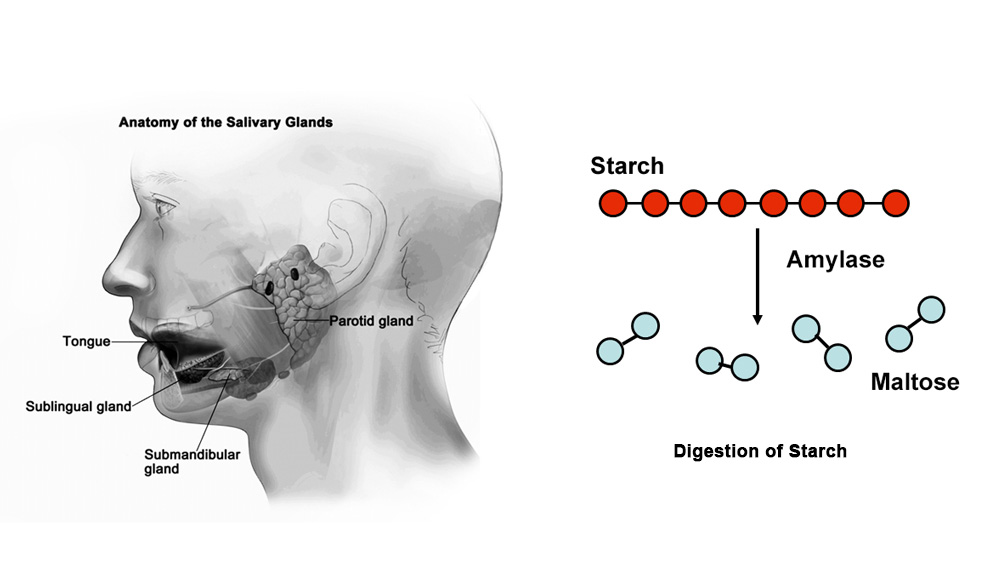
Akshi and Noor researched the Amylase and Starch reaction that occurs within our mouths. The enzyme amylase acts as a protein catalyst for biological reactions. Through this reaction, the starch will be hydrolyzed to glucose in varying amounts based on the time allowed for reaction. Iodine is used to test the presence or absence of starch in the solution by forming a blue-black complex with starch. The rate at which the blue-black colour complex disappears from solution, the faster the enzyme amylase is reacting with the starch, since Iodine does not react with glucose. The change in color will be detected by a light sensor and will stop the car.

Bhavya and Siddhant collaborated with the Laboratory supervisor, Mohammadkhalil Poush to recommend the titration of NaOH and CuSO4 for the Chem-E-Car stopping mechanism. NaOH would be titrated against CuSO4 with phenolphthalein as the indicator. The car would stop once a color change from red to light blue is detected. CuSO4 would be injected at a constant rate to NaOH, with CuSO4’s concentration depending on the distance the car has to travel. Unfortunately, this reaction has been placed on hold for the time being as it does not currently comply with the American Institute for Chemical Engineers (AICHE) regulations.
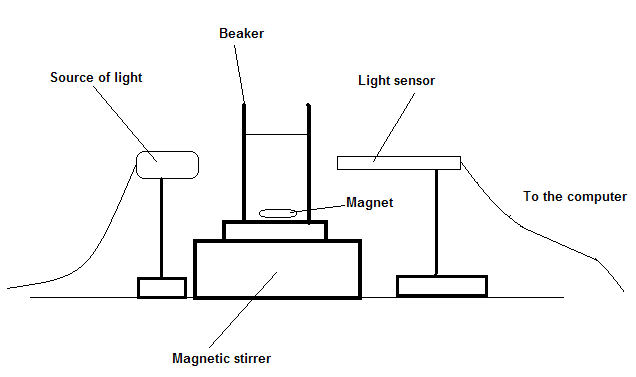
Abhay and Stephanie considered the Sulfur Clock reaction, in which the reactants, sodium thiosulfate and hydrochloric acid, are both colorless. When they are added in a beaker, sulfur is produced as a precipitate and it causes the solution to change from colorless to cloudy. A light source and a sensor is placed on opposing sides of the beaker, and the sensor will detect when the light has decreased beneath a threshold to activate the braking mechanism. This reaction is a first order rate reaction, and the reaction rate increases when sodium thiosulfate is increased.